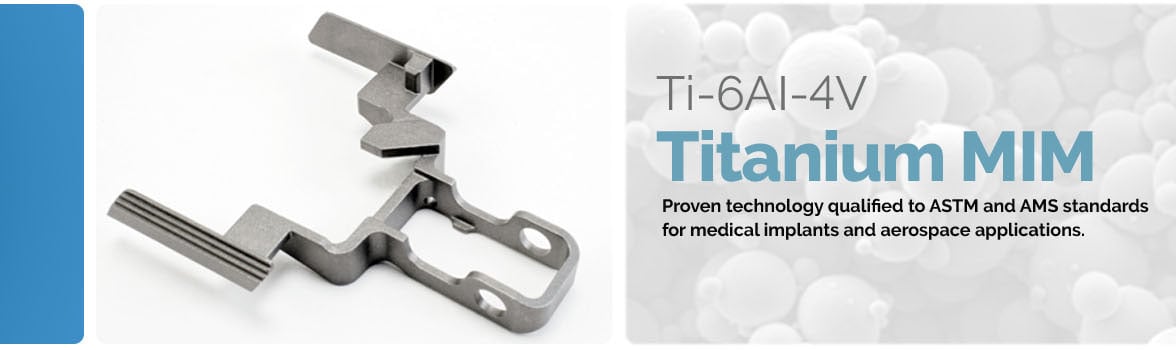
Praxis Powder Technology is an FDA-registered, ISO-13485-certified manufacturing company that produces titanium components via powder metallurgy for the medical, aerospace, consumer and sporting goods markets. Praxis specializes in porous titanium and titanium metal injection molding (MIM or TiMIM).
Praxis has perfected their proprietary process to provide high-performance titanium parts for a variety of demanding applications while satisfying stringent regulatory requirements. At Praxis, we recognize the importance of collaboration; from product development to regulatory submissions and supply chain management.
Device Master File (MAF)
In 2016, Praxis submitted a Device Master File to the FDA for their Ti-6Al-4V material to streamline customer product submissions. Guidance from ISO 10993 was utilized to conduct tests; please contact us for more details.